
Better Outcomes
Improved Lives™
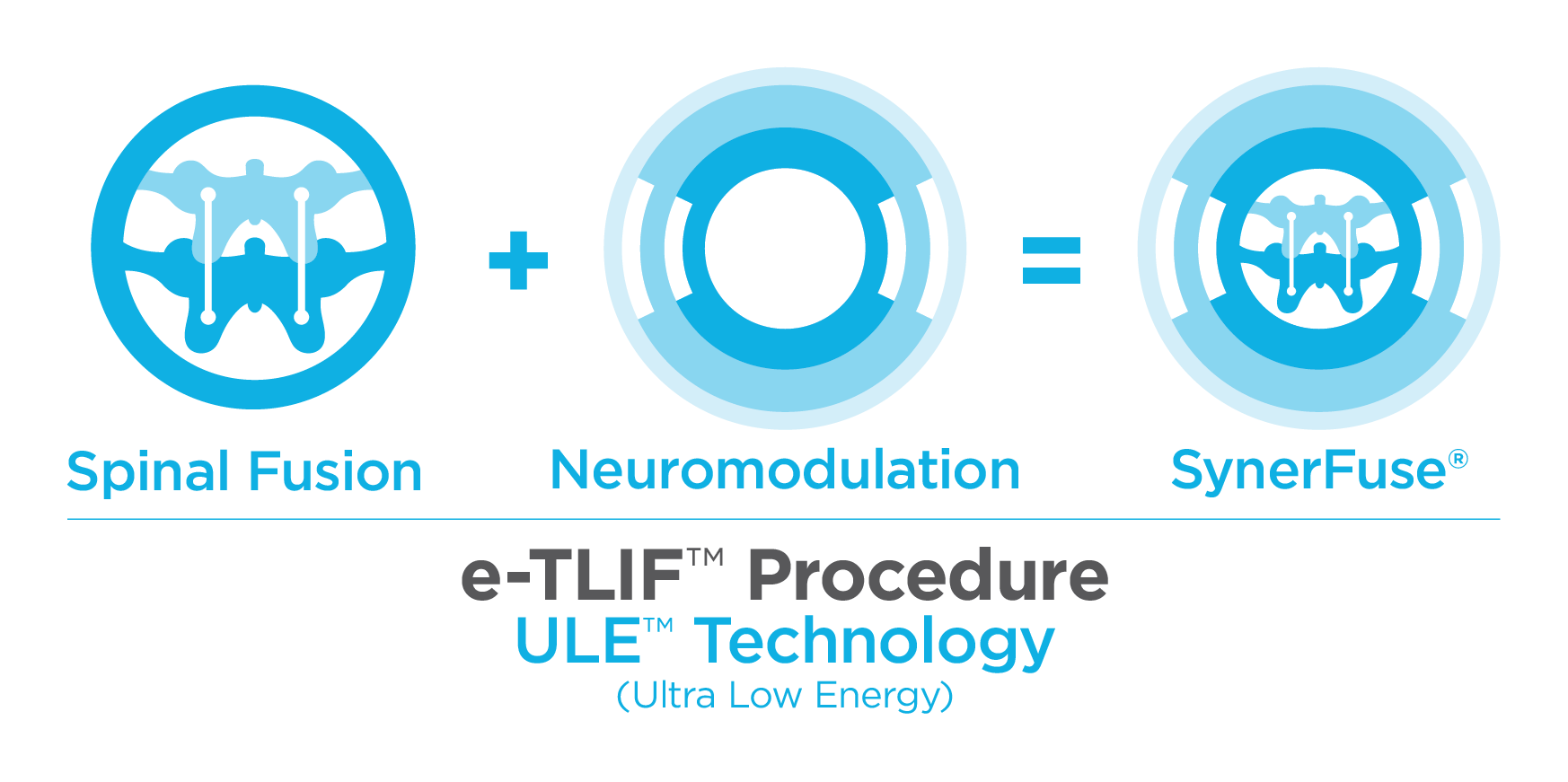
The SynerFuse® e-TLIF™ procedure, a ULE™ Therapy (Ultra Low Energy), is used to address chronic low back and leg pain for spinal fusion patients.
The Problem in the United States with Chronic Low Back Pain and Opioids

90 Million+
lives affected
1 Million+
back surgeries
100% of spinal fusion patients are prescribed opioids post-surgery.

100,000+
annual deaths
$1.5 Trillion
annual cost
40% of fusion patients have chronic pain, untreatable with opioids.

